Breakthrough!
by Lorien E. Menhennett
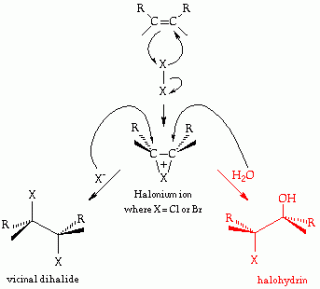 |
| This is an example of what I’ve been working on today … the reaction to the left is called halogenation; the reaction to the right is called halohydrin formation. |
After hours of wrestling through organic chemistry problems (and getting so many wrong) … breakthrough! I think I’m finally getting it. Reaction mechanisms, that is. Well, not ALL reaction mechanisms, just the handful that we’ve studied thus far this semester. (The mechanisms of SN1, SN2, E1, E2, hydrohalogenation, hydration, halogenation, halohydrin formation, and hydroboration-oxidation, to be more specific.)
This morning, when I started studying for my organic chemistry final, it was all a mish-mash to me. Anti addition, syn addition, retention of configuration, racemization, carbocations, bridged halonium ions … I couldn’t keep it all straight. But after making (and reviewing) a huge batch of flashcards and working through gobs of these practice problems, something has clicked. Just now, I found myself looking at a reaction and actually knowing what to do with the bonds, atoms, and electrons. What arrows to draw. What products (including stereoisomers!) would form.
I still have a lot of practicing to do. I don’t have all of these reactions down completely. But I’m getting the hang of it, slowly. Thankfully, my organic chemistry final isn’t until next Friday, so I have plenty of time to cement my understanding. But knowing that I am already on my way is a darn good feeling.