
There are some things that we just take for granted, it seems. The sky is blue, the grass is green, and the chemistry periodic table of elements looks something like the image at right.
But, as is the case with so many things, there are multiple ways of representing the information on the periodic table. There is a Web site called The Internet Database of Periodic Tables that I recently discovered, thanks to a dear friend who is a chemistry major, which has images of literally hundreds of periodic table representations. It fascinates me that there are so many ways of showing what we take for granted in chemistry class. Some of them are, perhaps, not so accurate anymore based on current information and data. Others are not really very scientific, and are more novelty items. Regardless, how amazing that people have taken the time (and you will see that many of these have REALLY taken time) to develop different ways of thinking about what our world is made of. Here are a few of my favorites. But I really encourage you, if you like any of what you see here, to visit the Web site and peruse the photos. You will laugh, scratch your head, and laugh again, I guarantee it.
Alternate views of the periodic table of elements:
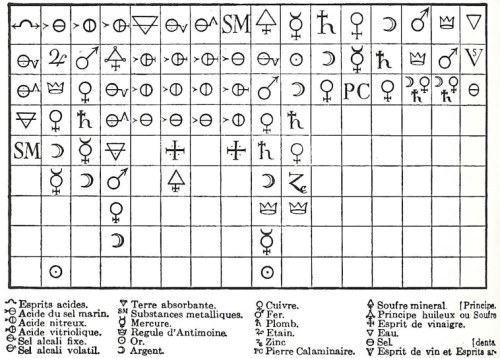
Étienne François Geoffroy’s 1718 Affinity Table. At the head of the column is a substance with which all the substances below can combine.
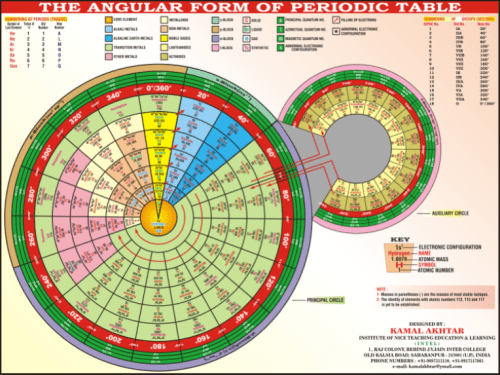
Angular Form of the Periodic Table by Kamal Akhtar: “The complete periodic table is consists of two circles, principal circle and auxiliary circle. The principal circle is consist of seven tracks (periods) and eighteen sectors (groups). The auxiliary circle is consist of only two tracks, inner track and outer track. There is no division of sectors in auxiliary circle.” (2008)
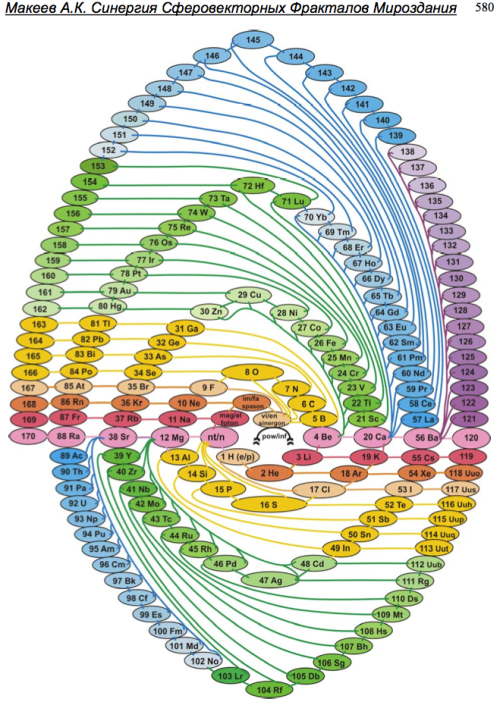
By Alexander Makeyev – integrated interdisciplinary researcher, inventor, poet (2011)
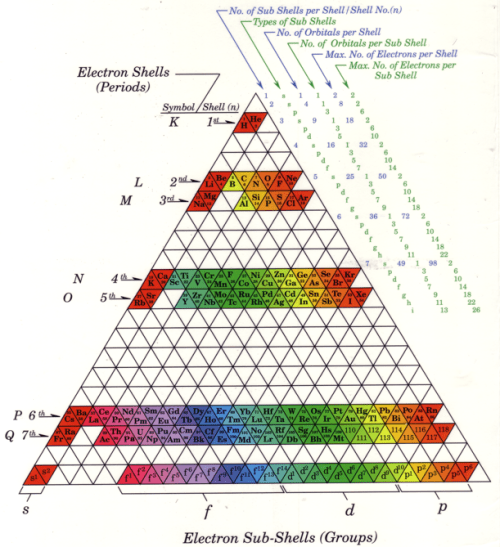
From Protege Publishing comes the Progressive Periodic Table: “The PPTE allows you to observe and learn the periodicity of the elements in a more aesthetically pleasing form. It removes confusion from the growth pattern between shell pairs, and solves the problem of keeping the lanthanides and actinides in sequence with the rest of the table.”

Bydgoszcz’s Periodic Table (2008).
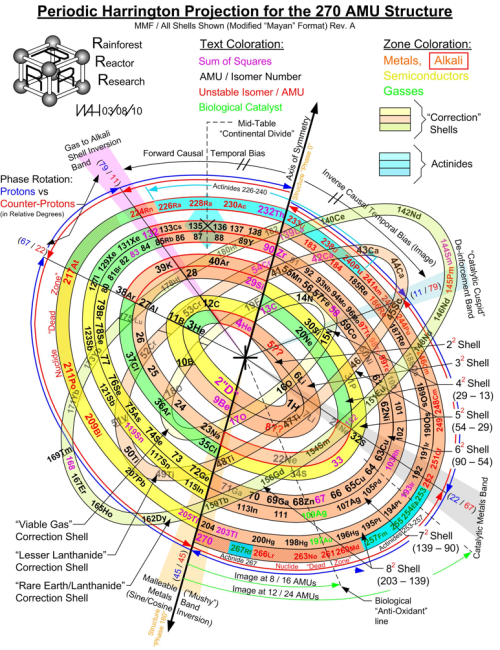
Harrington Projection for The 270 AMU Structure (2010).

Normal vs Correction Shell “Pi Paradox” for 1-270 AMUs.
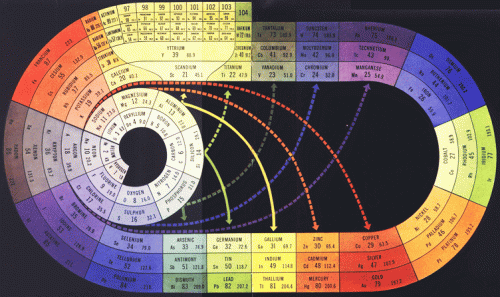
Origionally developed in 1933, the colour version of Clark’s arrangement is from Life Magazine, May 1949. This was the model for Longman’s 1951 mural.
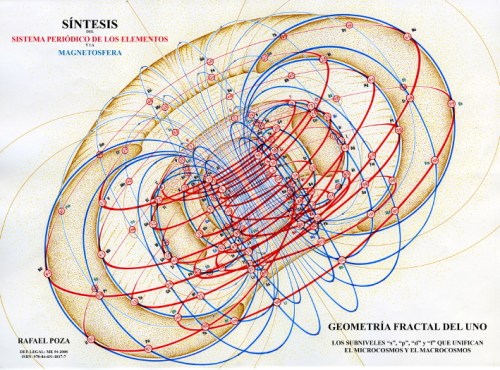
Rafael Poza’s Elements and the Magnetosphere (2008).
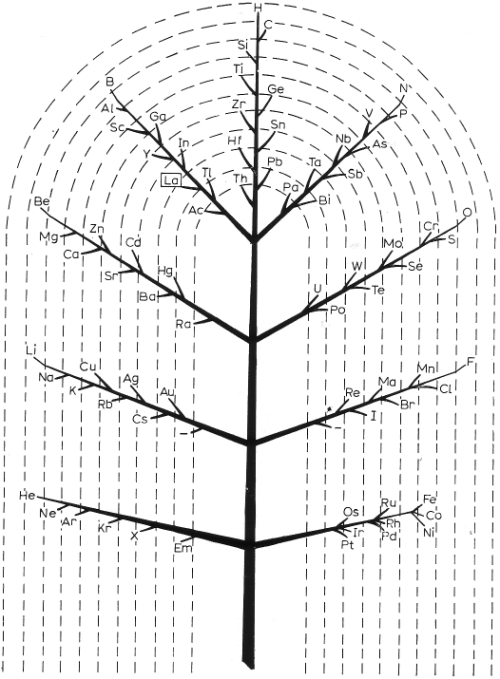
In 1944 Müller produced a formulation based on Darwin’s tree of life.
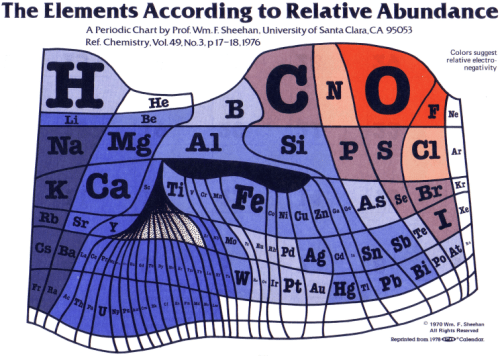
A 1970 periodic table by Prof. Wm. F. Sheehan of the University of Santa Clara that claims to show the elements according to relative abundance at the Earth’s surface.
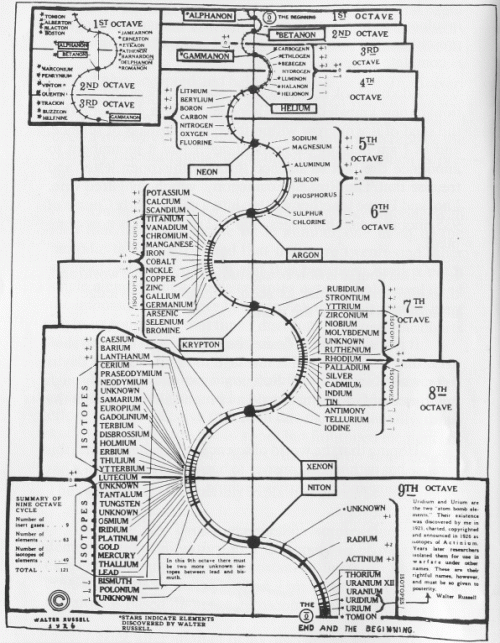
Walter Russell’s Periodic Chart of The Elements 1 (1926).
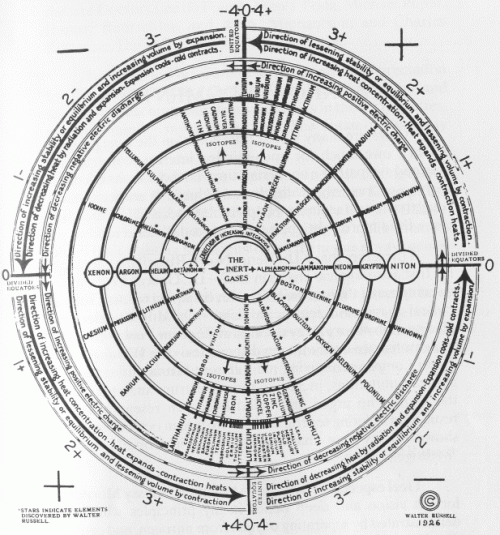
Walter Russell’s Periodic Chart of The Elements 2 (1926).
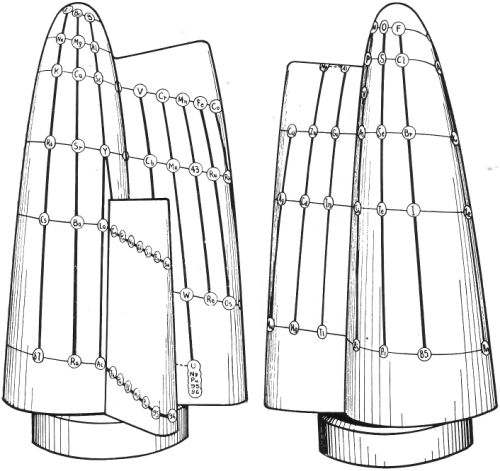
Stedman’s conic system of 1947.
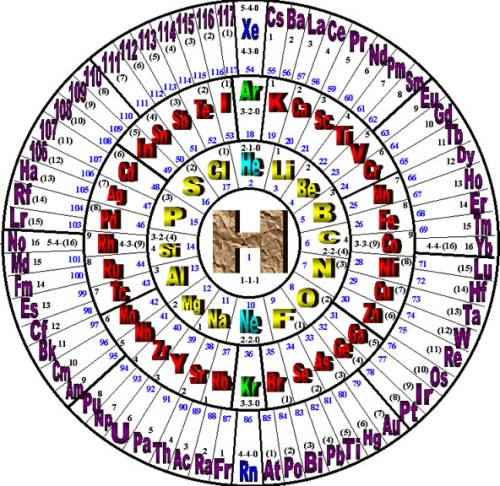
The Wheel of Motion (WoM) representation of the periodic table of elements shows the periodic nature of the elements, as developed in the Reciprocal System of Physical Theory (RST).
















